Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Seko, Noriaki*; Hoshina, Hiroyuki*; Kasai, Noboru*; Shibata, Takuya; Saiki, Seiichi*; Ueki, Yuji*
Radiation Physics and Chemistry, 143, p.33 - 37, 2018/02
Times Cited Count:16 Percentile:85.18(Chemistry, Physical)Chen, J.; Asano, Masaharu; Yamaki, Tetsuya; Yoshida, Masaru
Journal of Applied Polymer Science, 100(6), p.4565 - 4574, 2006/06
Times Cited Count:44 Percentile:75.8(Polymer Science)This study concerns a comparative study of three crosslinkers, divinylbenzene (DVB), 1,2-bis(p,p-vinylphenyl)ethane (BVPE) and triallyl cyanurate (TAC) crosslinked poly(ethylene-co-tetrafluoroethylene) (ETFE)-based radiation-grafted membranes, which were prepared by radiation grafting of p-methylstyrene (MeSt) onto ETFE films and subsequent sulfonation. The effect of the different types and contents of the crosslinkers on the grafting and sulfonation, and the properties such as water uptake, proton conductivity and thermal/chemical stability of the resulting polymer electrolyte membranes was investigated in detail. Introducing crosslink structure into the radiation-grafted membranes leads to a decrease in proton conductivity due to the decrease in water uptake. The thermal stability of the crosslinked radiation-grafted membranes is also somewhat lower than that of the noncrosslinked one. However, the crosslinked radiation-grafted membranes show significantly higher chemical stability characterized in the 3% H O
O at 50
at 50 C. Among the three crosslinkers, the DVB shows a most pronounced efficiency on the crosslinking of the radiation-grafted membranes, while the TAC has no significant influence; the BVPE is a mild and effective crosslinker, showing the moderate influence between the DVB and TAC crosslinkers.
C. Among the three crosslinkers, the DVB shows a most pronounced efficiency on the crosslinking of the radiation-grafted membranes, while the TAC has no significant influence; the BVPE is a mild and effective crosslinker, showing the moderate influence between the DVB and TAC crosslinkers.
 thin films by ion irradiation
thin films by ion irradiationNarumi, Kazumasa; Sakai, Seiji; Naramoto, Hiroshi*; Takanashi, Koki
JAEA-Review 2005-001, TIARA Annual Report 2004, p.238 - 240, 2006/01
no abstracts in English
 films polymerized by ion irradiation
films polymerized by ion irradiationNarumi, Kazumasa; Sakai, Seiji; Naramoto, Hiroshi*; Takanashi, Koki
Fullerenes, Nanotubes, and Carbon Nanostructures, 14(2-3), p.429 - 434, 2006/00
no abstracts in English
Sawada, Shinichi; Yamaki, Tetsuya; Asano, Masaharu; Terai, Takayuki*; Yoshida, Masaru
Transactions of the Materials Research Society of Japan, 30(4), p.943 - 946, 2005/12
We synthesized crosslinked-polytetrafluoroethylene (PTFE) electrolyte membranes by a radiation grafting technique under different conditions, and then investigated their proton conduction properties at controlled temperatures and relative humidities (R.H.) by an AC impedance method. The density and length of graft chains were controlled by varying the pre-irradiation dose and grafting time, respectively. When the pre-irradiation dose was fixed at 15 kGy to make the graft chains an uniform density, the elongation of the graft chain increased the ion exchange capacity (IEC), there by enhancing their proton conductivity. The membrane with an IEC of 2.8 meq/g possessed the maximum conductivity reaching 0.20 S/cm at 80  C and R.H. 95%. At almost the same IEC, membranes with more and shorter graft chains showed higher conductivity than those with less and longer chains. This result was probably related to the different structures of hydrophilic domains as proton-conducting pathways.
C and R.H. 95%. At almost the same IEC, membranes with more and shorter graft chains showed higher conductivity than those with less and longer chains. This result was probably related to the different structures of hydrophilic domains as proton-conducting pathways.
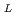 -proline methyl ester) gel membrane in LiCl solution
-proline methyl ester) gel membrane in LiCl solutionChen, J.; Asano, Masaharu; Tsubokawa, Norio*; Maekawa, Yasunari; Yamaki, Tetsuya; Yoshida, Masaru
Journal of Polymer Science, Part B; Polymer Physics, 43(20), p.2843 - 2851, 2005/10
Times Cited Count:0 Percentile:0.01(Polymer Science)Impedance spectra analysis of a thermo-responsive poly(acryloyl-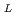 -proline methyl ester) (poly(A-ProOMe)) hydrogel membranes in an aqueous solution of LiCl was carried out using a simple equivalent model. The hydrogel membrane was synthesized by
-proline methyl ester) (poly(A-ProOMe)) hydrogel membranes in an aqueous solution of LiCl was carried out using a simple equivalent model. The hydrogel membrane was synthesized by  -radiation-induced polymerization and crosslinking of A-ProOMe monomer aqueous solution in a glass-cast. By means of the impedance spectra analysis, a novel method for the calculation of the ionic conductivity of the hydrogel membranes in LiCl solution was proposed. The calculated ionic conductivity was in agreement well with the determined value. In addition, effects of temperature and LiCl concentration on the impedance spectra and ionic conductivity of the gel membrane were analysized. Results indicated that the impedance spectra analysis is a very useful tool for evaluating the electric properties of gel membranes in an electrolyte solution. The poly(A-ProOMe) gel membrane in 1.0 M LiCl solution showed a high ionic conductivity of about 0.2 S/cm at 14
-radiation-induced polymerization and crosslinking of A-ProOMe monomer aqueous solution in a glass-cast. By means of the impedance spectra analysis, a novel method for the calculation of the ionic conductivity of the hydrogel membranes in LiCl solution was proposed. The calculated ionic conductivity was in agreement well with the determined value. In addition, effects of temperature and LiCl concentration on the impedance spectra and ionic conductivity of the gel membrane were analysized. Results indicated that the impedance spectra analysis is a very useful tool for evaluating the electric properties of gel membranes in an electrolyte solution. The poly(A-ProOMe) gel membrane in 1.0 M LiCl solution showed a high ionic conductivity of about 0.2 S/cm at 14 C. The temperature-dependence of the ionic conductivity was a complex nonlinear form due to the volume phase transition of the thermo-responsive poly(A-ProOMe) gel membrane, and the volume phase transition temperature appeared to be decreased with the increase in the LiCl concentration.
C. The temperature-dependence of the ionic conductivity was a complex nonlinear form due to the volume phase transition of the thermo-responsive poly(A-ProOMe) gel membrane, and the volume phase transition temperature appeared to be decreased with the increase in the LiCl concentration.
Enomoto, Kazuyuki*; Narita, Tadashi*; Maekawa, Yasunari; Yoshida, Masaru; Hamana, Hiroshi*
Journal of Fluorine Chemistry, 125(7), p.1153 - 1158, 2004/07
Times Cited Count:3 Percentile:10.94(Chemistry, Inorganic & Nuclear)Radical polyaddition of bis( -trifluoromethyl-
-trifluoromethyl- ,
, -difluorovinyl) terephthalate (BFP) with 1,4-dioxane (DOX) afforded higher molecular weight polymers under
-difluorovinyl) terephthalate (BFP) with 1,4-dioxane (DOX) afforded higher molecular weight polymers under  -rays compared to those yielded by benzoyl peroxide initiation. More detailed study on the radiation-induced polyaddition of BFP with DOX and optimization of the reaction conditions were carried out. It was necessary to irradiate with doses of 2000, 1500, and 750 kGy, to obtain quantitative conversion of BFP at the feed molar ratio DOX/BFP of 8.0, 16, and 32, respectively. Step-growth polymerization mechanism was suggested by the measurements of molecular weights of the polymers obtained with several irradiation doses. It was concluded that the molecular weight of the polymer could be controlled by the feed molar ratio of DOX/BFP and irradiation doses. The reaction between polymers might take place after the quantitative conversion of BFP. Radiation-induced radical polyaddition mechanism of BFP with DOX was proposed.
-rays compared to those yielded by benzoyl peroxide initiation. More detailed study on the radiation-induced polyaddition of BFP with DOX and optimization of the reaction conditions were carried out. It was necessary to irradiate with doses of 2000, 1500, and 750 kGy, to obtain quantitative conversion of BFP at the feed molar ratio DOX/BFP of 8.0, 16, and 32, respectively. Step-growth polymerization mechanism was suggested by the measurements of molecular weights of the polymers obtained with several irradiation doses. It was concluded that the molecular weight of the polymer could be controlled by the feed molar ratio of DOX/BFP and irradiation doses. The reaction between polymers might take place after the quantitative conversion of BFP. Radiation-induced radical polyaddition mechanism of BFP with DOX was proposed.
Zhaoxin, L.*; Fujimura, Takashi
Radiation Physics and Chemistry, 42(4-6), p.923 - 926, 1993/10
no abstracts in English
L.Z.Xin*; M.Carenza*; Kaetsu, Isao*; Kumakura, Minoru*; Yoshida, Masaru; Fujimura, Takashi
Radiation Physics and Chemistry, 40(6), p.579 - 584, 1992/00
no abstracts in English
JAERI-M 91-054, 44 Pages, 1991/03
no abstracts in English
*; Hosoi, Fumio; ; *
Chem.Pharm.Bull., 35(5), p.2045 - 2051, 1987/05
no abstracts in English
*; Hosoi, Fumio; ; *
Yakuzaigaku, 47(3), p.154 - 160, 1987/03
no abstracts in English
Fujimura, Takashi; Kaetsu, Isao
Biotechnol.Bioeng., 29, p.171 - 175, 1987/00
no abstracts in English
*; Hosoi, Fumio; ; *
Kobunshi Rombunshu, 43(1), p.51 - 58, 1986/00
Times Cited Count:2 Percentile:20.28(Polymer Science)no abstracts in English
*
JAERI-M 84-239, 59 Pages, 1985/01
no abstracts in English
Fujimura, Takashi; Kaetsu, Isao
Zeitschrift f r Naturforschung, C, 40, p.576 - 579, 1985/00
r Naturforschung, C, 40, p.576 - 579, 1985/00
no abstracts in English
*; ; ; *;
Journal of Polymer Science; Polymer Chemistry Edition, 22, p.3417 - 3421, 1984/00
no abstracts in English
*
JAERI-M 83-199, 83 Pages, 1983/11
no abstracts in English
Fujimura, Takashi; Kaetsu, Isao
Int.J.Appl.Radiat.Isot., 34(6), p.929 - 931, 1983/00
Times Cited Count:13 Percentile:91.35(Nuclear Science & Technology)no abstracts in English
Fujimura, Takashi; Kaetsu, Isao
Maku, 8(3), p.142 - 149, 1983/00
no abstracts in English